Self-ordering of pores into a two-dimensional hexagonal array is one of the unique properties of anodic aluminum oxide. For the first time the possibility of obtaining oxide films with a highly ordered arrangement of channels and an extremely narrow pore size distribution was demonstrated by Japanese scientists in 1995 [1]. This finding gave rise to a sharp increase in the number of publications on porous films of anodic aluminum oxide. However, over the next 20 years, the question why self-ordering of pores into a two-dimensional hexagonal array occurs only in a narrow voltage range remained open.
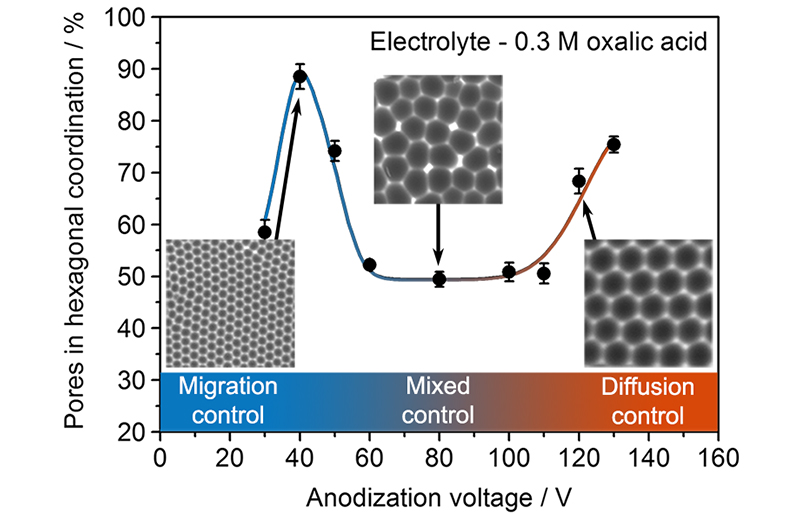
The researchers of our group have solved this riddle of Nature. Using the method of linear voltammetry, it was shown that the transition from a disordered to an ordered porous structure coincides with a change in the nature of a limiting stage.
A prerequisite for the formation of oxide films with an ordered arrangement of pores is carrying out the process in a regime where the growth rate of the oxide is limited by one of the following processes:
1) the migration of ions in the barrier layer or on its boundaries;
2) the diffusion of products and/or reagents in the channels of the nascent oxide film.
In this case, the mixed regime leads to the formation of disordered structure due to the constant branching of the channels, caused by their different growth rates. Universality and predictive power of the model was demonstrated for electrolytes based on oxalic and sulfuric acids of different concentrations. The obtained data open a way for a directed search of the conditions under which anodic aluminum oxide with a system of ordered channels is formed instead of the empirical retrieval currently used. More information can be found in our article published in the journal Electrochimica Acta [2].
[1] H. Masuda, K. Fukuda, Ordered Metal Nanohole Arrays Made by a Two-Step Replication of Honeycomb Structures of Anodic Alumina // Science, 268 (1995) 1466-1468. [2] I.V. Roslyakov, E.O. Gordeeva, K.S. Napolskii, Role of Electrode Reaction Kinetics in Self-Ordering of Porous Anodic Alumina // Electrochimica Acta, 241 (2017) 362-369.